Thread Song
Before we start , what is the randle cycle?
, It is a metabolic mechanism proposed by Philip Randle in 1963. which explains how muscle and adipoose tissues choose between burning Carbs or fats..
The randle cycle aka the glucose-fatty acid cycle is the reciprocal inhibtion between glucose oxidation and fatty-acid oxidation. High fatty acids shut down glucose use ,maily at PDH , and then PFK-1 and uptake). High glucose raises malonyl-CoA and blocks fatty-acid entry at CPT-I. It operates on top of insulin control , works in muscle , heart and liver , also has short-term(allosteric/phosphorylation) and long-term(gene expression) arms , and explains fuel switching in fastin, High-fat feeding, exercise , starvation and diabetes.
- History & Discovery
- Overview – How the Cycle Works Both Ways
- Fatty Acids Inhibiting Glucose Oxidation
- Glucose Inhibiting Fatty Acid Oxidation
- Short-Term Controls (Allosteric + Phosphorylation)
- Long-Term Controls (Gene Expression)
- Tissue-Specific Details & Physiological States
- Mitochondrial-Level Details
- AMPK Override Mechanism
- How the Randle Cycle Affects the Human Body: Impacts on BMI, Body Fat, and Skeletal Muscle
as i mentioned before , This cycle proposed in 1963 by Philip Randle.
Title: “The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus.”
They proved in an isolated rat heart and a diaphragm that fatty acids or ketones inhibt glucose uptake and oxidation independently of hormones.
in 1977 , MGarry & Foster added the reverse arm ( malonyl-CoA -> CPT-I)
In 2009, The definitive modern review: Hue and Taegtemyer.
The Randle cycle revisited a new head for an old hat.
These are the sources of Information about this part.
Title: “The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus.”
They proved in an isolated rat heart and a diaphragm that fatty acids or ketones inhibt glucose uptake and oxidation independently of hormones.
in 1977 , MGarry & Foster added the reverse arm ( malonyl-CoA -> CPT-I)
In 2009, The definitive modern review: Hue and Taegtemyer.
The Randle cycle revisited a new head for an old hat.
These are the sources of Information about this part.
Original 1963 Lancet paper: https://doi.org/10.1016/S0140-6736(63)91500-9
Hue & Taegtmeyer 2009 (free full text): https://pmc.ncbi.nlm.nih.gov/articles/PMC2739696/
Wikipedia overview + references: https://en.wikipedia.org/wiki/Randle_cycle
Randle 1998 review: https://pubmed.ncbi.nlm.nih.gov/10095997/
Hue & Taegtmeyer 2009 (free full text): https://pmc.ncbi.nlm.nih.gov/articles/PMC2739696/
Wikipedia overview + references: https://en.wikipedia.org/wiki/Randle_cycle
Randle 1998 review: https://pubmed.ncbi.nlm.nih.gov/10095997/
It is not a classic cycle but reciprocal regulation that matches the available fuel to the tissue's needs without making change in the total energy production.
High Circulating fatty acids aka lipolysis -> tissues burn fats->glucose is spared.
High glucose (the fed state) -> fat burning process is blocked->glucose is used or stored.
This fine-tunings sits on the top of the hormonal control.

High Circulating fatty acids aka lipolysis -> tissues burn fats->glucose is spared.
High glucose (the fed state) -> fat burning process is blocked->glucose is used or stored.
This fine-tunings sits on the top of the hormonal control.

β oxidation raises mitochondrial acetyl-CoA/CoA and NADH/NAD+ ratios
-> Direct inhibition of pyruvate dehydrogenase (PDH) complex [near the total block at high levels].
->Citrate builds up, exits to cytosol.
Cytosolic citrate:
• Inhibits PFK-1.
• Inhibits PFK-2 -> drops fructose-2,6-bisphosphate ( PFK-1’s strongest activator).
-Result: glucose-6-phosphate accumulates -> weaker hexokinase inhibition.
Order of inhibition strength: glucose uptake (20-30 %) < PFK-1 (40-60 %) < PDH (~100 %).
Spared glucose goes to glycogen or anaplerosis (pyruvate ->oxaloacetate).
-> Direct inhibition of pyruvate dehydrogenase (PDH) complex [near the total block at high levels].
->Citrate builds up, exits to cytosol.
Cytosolic citrate:
• Inhibits PFK-1.
• Inhibits PFK-2 -> drops fructose-2,6-bisphosphate ( PFK-1’s strongest activator).
-Result: glucose-6-phosphate accumulates -> weaker hexokinase inhibition.
Order of inhibition strength: glucose uptake (20-30 %) < PFK-1 (40-60 %) < PDH (~100 %).
Spared glucose goes to glycogen or anaplerosis (pyruvate ->oxaloacetate).
Glucose -> glycolysis -> citrate export -> acetyl-CoA carboxylase (ACC) Produces the malonyl-CoA.
The Malonyl-Coa allosterically inhibits CPT-I -> long-chain acyl-CoA cannot enter mitochondria.
Fatty acids are rerouted to triacylglycerol storage.
This is the McGarry/Foster arm that makes the system fully reciprocal.
The Malonyl-Coa allosterically inhibits CPT-I -> long-chain acyl-CoA cannot enter mitochondria.
Fatty acids are rerouted to triacylglycerol storage.
This is the McGarry/Foster arm that makes the system fully reciprocal.
- PDH: It is Inactivated by the PDK phosphorylation + allosteric inhibition (acetyl-CoA, NADH); activated by the PDP dephosphorylation (Ca^2+-sensitive).
- PFK-1 / PFK-2: Citrate inhibition.
- ACC: It is Activated by citrare , inactivated by AMPL at Ser79 (ACC2).
- Malonyl-CoA decarboxylase (MCD): It is activated bt contraction.
- CPT-I: Malonyl-CoA sensivitity is higher in muscle than liver.
- Liver glucokinase: It is inhbited by long-chain acyl-CoA.
- PDK4 is upregulated by fasting/High-fat via PPARα -> locks the PDH Off.
- PPARα upregulates FAO Gense , CPT-I, MCD ( the liver/heart)
- SREBP-1c and ChREBP ( Glucose-driven) upregulate lipogenic enzynes in liver.
- PPARγ in adipose promotes storage.
- Heart and oxidative muscles: Classic Randle effect strongest (GLUT4-dependent , no glucokinase).
- Liver: Glucokinase + Ketogenesis link.
- Fasted State: High FA/Ketones -> PDH is Off -> pyrubate/lacate spared for gluconeogenesis + the brain.
- Exercising: explains rapid post-exercies glycogen resynthesis.
- Starvation/Diabetes: High muscle glycogen despire impaired glucose oxidation.
Fatty-acid oxidation raises the NADH/NAD+ , and Mitochondrial Membrane potential , and shifts electrons toward complex II. Loweres the ATP/O ratio and Highers the ROS potential compared with the glucose oxidation.
High mitochondrial membrane potential favors fatty acids , meanwhile low mitochondrial membrane potential favors glucose
High mitochondrial membrane potential favors fatty acids , meanwhile low mitochondrial membrane potential favors glucose
Energy stess ( like exercise , ischemia) -> AMP rises -> AMPK activates.
Phosphorylates and inactivates AAC -> Malonyl-CoA drop -> Fatty-acid oxidation is allowed.
Activates PFK-2 -> Fructose-2,6-BP rises -> glycolysis up.
Stimulates the GLUT4 Translocation.
Completeley overrides the fatty-acid inhibition when energy demand spikes ( for example . Protects the heart during ischemia).
Phosphorylates and inactivates AAC -> Malonyl-CoA drop -> Fatty-acid oxidation is allowed.
Activates PFK-2 -> Fructose-2,6-BP rises -> glycolysis up.
Stimulates the GLUT4 Translocation.
Completeley overrides the fatty-acid inhibition when energy demand spikes ( for example . Protects the heart during ischemia).
Normal Physiology (Healthy Lean Individuals)
Metabolic Flexibility: When plasma free fatty acids (FFA) rise (on Fasting ,High-fat meal , or exercise recovery) , β-oxidation raises acetyl-CoA and citrate -> classic Randle inhibition of PDH > PFK-1 > GLUT4/Hexokinase. Muslce spares glucose (For brain / red-blood cells) and burns fats instead.
Reverse Arm: Post-meal glucose surge raises malonyl-CoA-> CPT-I bloc -> fat is stored rather than oxidized.
Net Effect on the body: Maintains stable blood glucose during the intermittent feeding ; prevents the wasteful simulatneuos oxidation of both fuels; Keeps total energy production constan while matching fuel to availability. This is exactly why lean people switch between carb and fat buring without blood-sugar spikes or crahses.
In Obesity and Elevated BMI (BMI ≥ 30 kg/m^2)
Obese individuals have chronically elevated circulating FFA (often 2x higher than lean controls) , because the enlarged adipose tissue + impaired insulin suppression of lipolysis floods the system. The Randle cycle is then presistently activated in muscles:
Direct inhibition of glucoes use: The PDH near total block + PFK-1 inhibition -> glucose 6 phosphate accumulates -> reduced glucose uptake and oxidation.
Result: Muscle insulin resistance.
This is the origianl Randle Explanation for why obese people shows impaired carb tolerance even before full type-2 diabetes develops.
Quantitative impact: Muscle glucose disposal drops significantly; whole-body insulin sensitivity falls because muscle is the main glucose sink. Compensatory hyperinsulinemia then drives further fat storage in adipocytes, raising BMI further (vicious cycle).
Incomplete fat oxidation and IMCL buildup: Many obese muscles also show reduced mitochondrial capacity (lower CPT-I, β-HAD, citrate synthase activity; fewer type-I oxidative fibers; smaller/fragmented mitochondria). FFA influx exceeds oxidation capacity → accumulation of intramyocellular lipids (IMCL). While IMCL itself is not always toxic (see “athlete’s paradox”), the lipid intermediates (diacylglycerol, long-chain acyl-CoAs, ceramides) activate PKCθ → serine phosphorylation of IRS-1 → blocked PI3K/Akt → further GLUT4 translocation failure. This amplifies the insulin resistance started by the classic Randle cycle.Studies show:
Extremely obese (BMI ~38 kg/m²) have 58–83 % lower muscle fatty-acid oxidation rates than lean (BMI ~24 kg/m²).
Negative correlation between IMCL content (measured by ¹H-MRS) and insulin sensitivity (r values often –0.7 to –0.95).


Direct Effects on Body Fat Mass and Distribution
Fat oxidation promoted: In calorie deficit or fasting, high FFA + active Randle drives muscle fat burning -> Help mobilize and burn stored triglycerides, aiding fat loss.
Fat storage promoted (chronic state): The reverse arm (high glucose -> malonyl-CoA) plus insulin resistance hyperinsulinemia keeps fat in adipocytes and promotes ectopic deposition in muscle and liver. This shifts body composition toward higher fat mass percentage and worse visceral/ectopic distribution — exactly what raises BMI and metabolic risk.
Direct Effects on Muscle
Fuel preference shift: Muscle becomes “fat-adapted” but glucose-intolerant ->lower glycogen synthesis efficiency post-meal, potential for reduced exercise performance in mixed-fuel activities.
Preservation vs. dysfunction: In starvation, the cycle spares muscle protein (less need for gluconeogenesis from amino acids). In chronic obesity, the combination of Randle + IMCL intermediates contributes to mitochondrial stress, higher ROS, and eventual sarcopenic obesity in severe cases.
Exercise override: AMPK activation during contraction drops malonyl-CoA and activates PFK-2 ->completely overrides Randle inhibition, explaining why exercise rapidly restores insulin sensitivity and glycogen resynthesis even in obese individuals.
Overall Body-Wide Consequences
Contributes to the metabolic syndrome cluster (insulin resistance, hyperglycemia, dyslipidemia) that accompanies high BMI.
Explains part of why weight loss (reduced FFA and IMCL) rapidly improves insulin sensitivity even before major fat mass changes.
In heart muscle (parallel effects): chronic Randle shift to fat oxidation reduces cardiac efficiency and can promote lipotoxicity — but the primary whole-body driver is skeletal muscle. Fgure representing the whole thing:
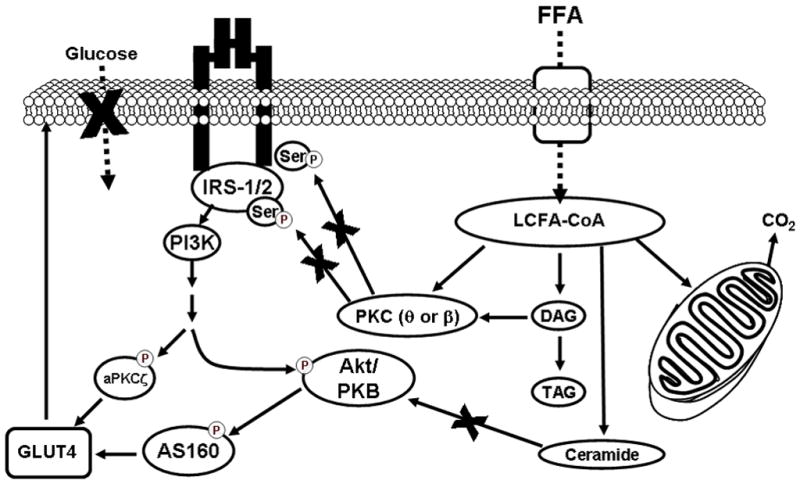
Metabolic Flexibility: When plasma free fatty acids (FFA) rise (on Fasting ,High-fat meal , or exercise recovery) , β-oxidation raises acetyl-CoA and citrate -> classic Randle inhibition of PDH > PFK-1 > GLUT4/Hexokinase. Muslce spares glucose (For brain / red-blood cells) and burns fats instead.
Reverse Arm: Post-meal glucose surge raises malonyl-CoA-> CPT-I bloc -> fat is stored rather than oxidized.
Net Effect on the body: Maintains stable blood glucose during the intermittent feeding ; prevents the wasteful simulatneuos oxidation of both fuels; Keeps total energy production constan while matching fuel to availability. This is exactly why lean people switch between carb and fat buring without blood-sugar spikes or crahses.
In Obesity and Elevated BMI (BMI ≥ 30 kg/m^2)
Obese individuals have chronically elevated circulating FFA (often 2x higher than lean controls) , because the enlarged adipose tissue + impaired insulin suppression of lipolysis floods the system. The Randle cycle is then presistently activated in muscles:
Direct inhibition of glucoes use: The PDH near total block + PFK-1 inhibition -> glucose 6 phosphate accumulates -> reduced glucose uptake and oxidation.
Result: Muscle insulin resistance.
This is the origianl Randle Explanation for why obese people shows impaired carb tolerance even before full type-2 diabetes develops.
Quantitative impact: Muscle glucose disposal drops significantly; whole-body insulin sensitivity falls because muscle is the main glucose sink. Compensatory hyperinsulinemia then drives further fat storage in adipocytes, raising BMI further (vicious cycle).
Incomplete fat oxidation and IMCL buildup: Many obese muscles also show reduced mitochondrial capacity (lower CPT-I, β-HAD, citrate synthase activity; fewer type-I oxidative fibers; smaller/fragmented mitochondria). FFA influx exceeds oxidation capacity → accumulation of intramyocellular lipids (IMCL). While IMCL itself is not always toxic (see “athlete’s paradox”), the lipid intermediates (diacylglycerol, long-chain acyl-CoAs, ceramides) activate PKCθ → serine phosphorylation of IRS-1 → blocked PI3K/Akt → further GLUT4 translocation failure. This amplifies the insulin resistance started by the classic Randle cycle.Studies show:
Extremely obese (BMI ~38 kg/m²) have 58–83 % lower muscle fatty-acid oxidation rates than lean (BMI ~24 kg/m²).
Negative correlation between IMCL content (measured by ¹H-MRS) and insulin sensitivity (r values often –0.7 to –0.95).


Direct Effects on Body Fat Mass and Distribution
Fat oxidation promoted: In calorie deficit or fasting, high FFA + active Randle drives muscle fat burning -> Help mobilize and burn stored triglycerides, aiding fat loss.
Fat storage promoted (chronic state): The reverse arm (high glucose -> malonyl-CoA) plus insulin resistance hyperinsulinemia keeps fat in adipocytes and promotes ectopic deposition in muscle and liver. This shifts body composition toward higher fat mass percentage and worse visceral/ectopic distribution — exactly what raises BMI and metabolic risk.
Direct Effects on Muscle
Fuel preference shift: Muscle becomes “fat-adapted” but glucose-intolerant ->lower glycogen synthesis efficiency post-meal, potential for reduced exercise performance in mixed-fuel activities.
Preservation vs. dysfunction: In starvation, the cycle spares muscle protein (less need for gluconeogenesis from amino acids). In chronic obesity, the combination of Randle + IMCL intermediates contributes to mitochondrial stress, higher ROS, and eventual sarcopenic obesity in severe cases.
Exercise override: AMPK activation during contraction drops malonyl-CoA and activates PFK-2 ->completely overrides Randle inhibition, explaining why exercise rapidly restores insulin sensitivity and glycogen resynthesis even in obese individuals.
Overall Body-Wide Consequences
Contributes to the metabolic syndrome cluster (insulin resistance, hyperglycemia, dyslipidemia) that accompanies high BMI.
Explains part of why weight loss (reduced FFA and IMCL) rapidly improves insulin sensitivity even before major fat mass changes.
In heart muscle (parallel effects): chronic Randle shift to fat oxidation reduces cardiac efficiency and can promote lipotoxicity — but the primary whole-body driver is skeletal muscle. Fgure representing the whole thing:

If you read till the end ,i really appreciate how you took time reading it.
that's it, Explained the Randle cycle and its effects in the human body.
Feel free to ask any question, Also you can correct me if there any misinformation in this article.
Yeah ,i call myself autistic for a reason......

Hue & Taegtmeyer 2009 (best review + the diagram above): https://pmc.ncbi.nlm.nih.gov/articles/PMC2739696/
Original 1963 paper: https://doi.org/10.1016/S0140-6736(63)91500-9
Original 1963 paper: https://doi.org/10.1016/S0140-6736(63)91500-9



